This article was researched and reviewed by Leo, an indoor plant specialist.
Bare-rooting is a management technique used when a plant’s root system has been compromised by fungal pathogens. For example, a *Philodendron billietiae* variegated that has lost its root system to a localized fungal bloom must be managed without substrate to prevent further decay. In these instances, the plant is maintained without perlite or soil while the root system is assessed and treated.
Guide Overview
- The Bare-Root Countdown: Duration of Viability
- The 24-Hour Critical Window for Exposed Roots
- The 7-Day Buffer: Survival in High-Humidity Environments
- Aroids vs. Succulents: Species-Specific Survival Rates
- Signs of Fatal Desiccation: Identifying Irreversible Damage
- Substrate Removal: A Method for Eradicating Fungus Gnats
- Breaking the Fungus Gnat Life Cycle
- Eliminating Soil-Borne Pathogens and Larvae
- Identifying Total Substrate Failure
- The Practical Advantages of Sterile Environments
- The Oxygen Paradox: Gas Exchange in Water vs. Soil
- Root Respiration and Gas Exchange Requirements
- Preventing Anaerobic Bacteria in Temporary Water Baths
- The Importance of Water Aeration for Bare Roots
- Hypoxia Symptoms: Identifying Oxygen Depletion
- Managing Shock: Preventing Post-Substrate Collapse
- The Role of Relative Humidity in Root Recovery
- Managing Foliar Wilt After Substrate Removal
- Rebuilding Mycorrhizal Networks in New Media
- Using Humidity Domes to Maintain Hydraulic Pressure
- Transitioning to Permanent Hydroponic Growth
- Identifying Nutrient Deficiencies and Chlorosis
- Species Suitable for Permanent Water Culture
- Selecting Appropriate Hydroponic Nutrients
- Removing Algae and Biofilm from Exposed Roots
- The Bare-Root Protocol: A Step-by-Step Procedure
- Substrate Removal: The Lukewarm Rinse Method
- Root Trimming for Rot Prevention
- The Hydrogen Peroxide Bath: Sterilization Procedures
- The Temporary Holding Tank Configuration
Bare-rooting is a technical procedure used for emergency intervention, plant transport, or transitioning to different growth media. When a plant is removed from its substrate, it enters a state of reduced metabolic activity. This process is necessary when shipping plants across long distances, managing severe infestations of fungus gnats, or moving specimens into semi-hydroponic systems.

The Bare-Root Countdown: Duration of Viability
Removing a plant from soil eliminates its primary moisture reservoir, thermal buffer, and structural support. Once roots are exposed to air, the plant’s survival depends on vascular pressure and cellular integrity. The timeframe for viability is determined by environmental conditions and species-specific physiology.
The 24-Hour Critical Window for Exposed Roots
During the initial 24 hours of exposure, root hairs are the most susceptible to damage. These microscopic, single-cell outgrowths are the primary sites for water and nutrient absorption. Unlike adventitious roots, root hairs lack protective structures against desiccation.
In low-humidity environments, root hairs can desiccate within 2 to 4 hours. This loss compromises the plant’s ability to absorb water, even upon reintroduction to a growth medium. Symptoms include a loss of turgor pressure, characterized by soft leaves and drooping petioles. If a plant remains bare-rooted for 24 hours without protection, it must typically regenerate its fine root system to resume normal function.
The 7-Day Buffer: Survival in High-Humidity Environments
Ambient humidity levels above 80% significantly extend the survival window of bare-rooted plants, often up to 7 days. High humidity reduces the rate of transpiration, which is the loss of water through the foliage.
For example, *Anthurium* specimens can be maintained in a sealed container with damp moss for approximately one week. The saturated air reduces the moisture gradient between the leaf and the environment, slowing internal water loss. Beyond seven days, the risk of opportunistic fungal and bacterial growth increases due to stagnant air conditions.
Aroids vs. Succulents: Species-Specific Survival Rates
Plant species vary in their tolerance for soil-less conditions. Aroids possess fleshy roots that provide limited internal water storage, allowing *Philodendron* species to survive several days if kept moist. However, their metabolic rates eventually deplete these reserves.
Succulents and cacti exhibit high tolerance for bare-rooting. Genera such as *Echeveria* and *Haworthia* can remain viable for up to two months without substrate. These plants utilize Crassulacean Acid Metabolism (CAM), opening stomata only at night to conserve water. Their root systems are evolutionarily adapted to prolonged drought. Bare-rooting is a standard procedure for treating root rot in succulents, as detailed Guide Overview on saving overwatered snake plants.
Signs of Fatal Desiccation: Identifying Irreversible Damage
Irreversible damage is indicated by changes in the vascular bundles. Roots that have turned grayish-black and become brittle have suffered cellular death. These tissues cannot be revived.
Foliar indicators include rapid marginal desiccation, resulting in papery leaf edges. If the primary stem or rhizome exhibits significant shriveling, internal hydraulic pressure has failed. In these cases, propagation from the remaining viable tissue is the only remaining option for preservation.
Substrate Removal: A Method for Eradicating Fungus Gnats
When standard pest control methods such as sticky traps or neem oil drenches fail, total substrate removal is a viable technical solution. This process eliminates the environment supporting the infestation.
Breaking the Fungus Gnat Life Cycle
Fungus gnats (*Bradysia* spp.) thrive in decaying organic matter and consistent moisture. Larvae inhabit the upper 2-3 inches of soil, where they consume fungi and root hairs.
Bare-rooting serves as an ecological reset by removing the breeding site. The absence of soil eliminates the habitat for eggs, larvae, and pupae. This is considered a definitive fungus gnat control measure. Thoroughly washing the roots ensures that no larvae are transferred to the new growth medium.
Eliminating Soil-Borne Pathogens and Larvae
Substrate can harbor pathogens such as *Pythium*, *Rhizoctonia*, and *Phytophthora*, which cause root rot. These organisms proliferate in the water film between soil particles.
For example, persistent fungal infections in *Monstera adansonii* often require bare-rooting, followed by a soak in a fungicide or hydrogen peroxide solution, before transitioning to a sterile medium like LECA. This allows for direct inspection and treatment of the root system, which is not possible when the plant is potted.
Identifying Total Substrate Failure
Substrate failure occurs when organic components, such as peat or coco coir, decompose into an anaerobic sludge. This material obstructs macro-pores, preventing gas exchange and suffocating the roots.
Substrate that emits a sulfurous odor indicates the presence of sulfur-reducing bacteria. In this state, the medium is toxic to the plant. The technical solution is to strip the roots, discard the failed substrate, and restart the plant in fresh media.
The Practical Advantages of Sterile Environments
Maintaining plants in water culture provides visual transparency, allowing for real-time monitoring of root health. This removes the uncertainty associated with soil moisture levels. For many growers, this transparency simplifies the management of plant health and reduces the complexity of care routines.

The Oxygen Paradox: Gas Exchange in Water vs. Soil
The ability of a plant to survive in pure water while failing in overwatered soil is explained by fluid dynamics and dissolved oxygen levels.
Root Respiration and Gas Exchange Requirements
Roots require oxygen for cellular respiration. In healthy soil, macro-pores facilitate gas exchange. Overwatering fills these pores, depleting available oxygen. High metabolic rates, often driven by intense grow lighting, increase the plant’s oxygen demand.
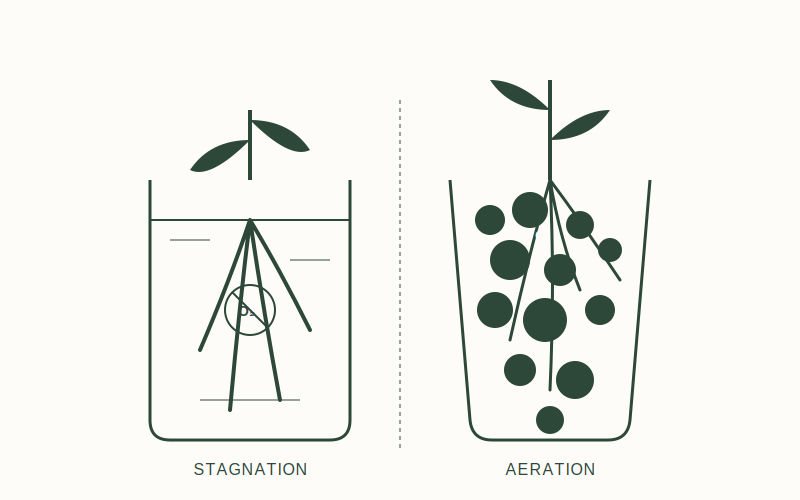
Preventing Anaerobic Bacteria in Temporary Water Baths
In a water-filled container, oxygen enters through surface diffusion. However, diffusion rates are slow, and oxygen levels at the bottom of a container can reach zero, creating an anaerobic environment.
Anaerobic bacteria produce biofilm as a byproduct in these low-oxygen conditions. To prevent this, water should be replaced every 48 hours to maintain dissolved oxygen levels and remove metabolic waste.
The Importance of Water Aeration for Bare Roots
For long-term water culture, mechanical aeration is required. Using an air stone and pump maintains dissolved oxygen levels at approximately 8-9 mg/L at room temperature. This inhibits the growth of anaerobic pathogens and supports root metabolism.
Hypoxia Symptoms: Identifying Oxygen Depletion
Wilting in a water-rich environment is a symptom of hypoxia. Insufficient oxygen prevents the molecular pumps in the roots from transporting water into the xylem.
If roots become translucent or mushy, cellular membranes have ruptured due to a lack of adenosine triphosphate (ATP). This loss of membrane integrity results in the death of the root tissue.
Managing Shock: Preventing Post-Substrate Collapse
Removing a plant from its substrate is a significant physiological stressor. Soil provides a stable microbial environment and consistent moisture, the loss of which requires careful management.
The Role of Relative Humidity in Root Recovery
Managing the Vapor Pressure Deficit (VPD) is critical during recovery. VPD is the difference between the moisture content of the air and its saturation point.
A low VPD is preferred for bare-rooted plants to minimize transpiration. This can be achieved using humidifiers or by creating a localized micro-climate with clear plastic enclosures. These methods reduce the hydraulic load on the roots during the adjustment period.
Managing Foliar Wilt After Substrate Removal
Wilting is a physiological response to reduce the leaf surface area exposed to light and air, thereby slowing water loss. If wilting occurs, the plant should be moved to an area with lower light intensity to reduce photosynthetic water demand. Foliar misting can also provide temporary cooling and limited hydration through the stomata.
Rebuilding Mycorrhizal Networks in New Media
Bare-rooting disrupts the symbiotic relationship between roots and mycorrhizal fungi. When transitioning to a new medium, such as LECA or fresh soil, the application of a mycorrhizal inoculant can assist in re-establishing these networks. Observations indicate that inoculated plants recover from bare-rooting stress approximately 30% faster than untreated plants.
Using Humidity Domes to Maintain Hydraulic Pressure
Humidity domes are used for smaller specimens to maintain internal hydraulic pressure. This prevents cavitation or embolisms within the xylem, which are permanent blockages caused by the breaking of the water column. Domes ensure that transpiration does not exceed the plant’s current water uptake capacity.
Transitioning to Permanent Hydroponic Growth
Transitioning to permanent water culture or semi-hydroponics is a method for maintaining a sterile growth environment and simplifying nutrient management.
Identifying Nutrient Deficiencies and Chlorosis
Tap water lacks essential macronutrients (N-P-K). Prolonged exposure to plain water results in chlorosis, or the yellowing of leaves, as the plant translocates nitrogen from older tissues to support new growth.
Successful water culture requires a complete hydroponic nutrient solution that includes micronutrients such as molybdenum and boron. These are often absent from standard soil fertilizers.
Species Suitable for Permanent Water Culture
Species suitability for water culture varies:
- High Suitability: *Aglaonema*, *Spathiphyllum*, *Philodendron hederaceum*, and *Epipremnum aureum*. These species adapt well to aqueous environments.
- Moderate Suitability: *Monstera*, *Syngonium*, and *Alocasia*. These require high levels of water aeration to prevent rhizome rot.
- Low Suitability: *Ficus lyrata*, most succulents, and woody-stemmed plants. These are prone to stem rot in permanent water culture.
Selecting Appropriate Hydroponic Nutrients
Three-part nutrient systems allow for the adjustment of N-P-K ratios according to the plant’s growth stage. High-phosphorus formulas are used initially to stimulate the development of water-adapted roots.
Nutrient concentrations should be managed carefully; plants in water are more susceptible to fertilizer burn. Initial applications should be at 25% of the recommended strength. If foliar tip necrosis occurs, the roots should be flushed with distilled water to remove excess salts.
Removing Algae and Biofilm from Exposed Roots
Algae competes for nutrients and oxygen. Its growth is stimulated by light exposure in water vessels.
To prevent algae, use opaque containers. Existing algae can be removed by gently wiping the roots and rinsing them in a dilute hydrogen peroxide solution (1 tablespoon of 3% H2O2 per gallon of water).
The Bare-Root Protocol: A Step-by-Step Procedure
This protocol outlines the technical steps for removing a plant from substrate and preparing it for recovery.
Substrate Removal: The Lukewarm Rinse Method
Avoid mechanical shaking, which can damage secondary roots. Use lukewarm water to rinse the substrate away; cold water can cause vascular contraction and stomatal closure.
Gently massage the root ball under running water until approximately 95% of the substrate is removed. Attempting to achieve total sterility often causes excessive root damage. If soil organisms are present, they will be removed during this process.
Root Trimming for Rot Prevention
Inspect the roots for signs of decay (black, mushy tissue or foul odors). Use sterilized shears to remove compromised tissue, cutting back to healthy, white sections.
If more than 50% of the root system is removed, reduce the foliage by approximately 30%. This balances the plant’s transpirational demand with its reduced uptake capacity.
The Hydrogen Peroxide Bath: Sterilization Procedures
Submerge the roots in a solution of 1 part 3% hydrogen peroxide to 3 parts water for 10-15 minutes. This oxidizes organic matter and pathogens, reducing the risk of re-infection. The process also increases oxygen availability to the root tissues.
The Temporary Holding Tank Configuration
Place the cleaned plant in a clear glass container filled with distilled water and a small amount of seaweed extract. Seaweed extract contains auxins and cytokinins that promote root development.
The container should be placed in a warm area with bright, indirect light. Monitor the roots daily for the emergence of new adventitious roots. Once these reach one inch in length, the plant can be transitioned to its final growth medium.
Successful bare-rooting requires adherence to principles of plant physiology, including the management of oxygen levels, humidity, and hydraulic pressure. By maintaining these variables, plants can be successfully transitioned or recovered from substrate-related issues.
Maintain consistent water changes and monitor environmental conditions to ensure long-term viability.