# Methods for Propagating and Gifting Spider Plant Offsets
*This article was researched and reviewed by Leo, an indoor plant specialist.*
In environments with low humidity, such as those utilizing forced-air heating, *Chlorophytum comosum* (spider plant) frequently produces offsets. These offsets, or plantlets, require specific propagation techniques to ensure a high survival rate when transitioning to independent growth.
Guide Overview
- The Anatomy of a Spiderette: When to Cut and When to Wait
- The Role of Node Development
- The Propagation Protocol: Water, Soil, or Pinning?
- Hydroponic Rooting
- Direct Soil Planting
- The Pinning Method
- The Root-to-Soil Transition
- Oxygenation and Soil Structure
- Pest Quarantine: Preventing Thrips Infestations
- The Smear Test for Pest Detection
- Gifting Protocols and Pest Prevention
- Terracotta for New Plantlets
- Care Documentation
- Managing Plant Density in Limited Spaces
- Nutrient Diversion and Mother Plant Health
- Vertical Space Management
Proper management of spider plant offsets involves understanding the biological connection between the parent plant and the plantlet. Improper removal can lead to high mortality rates and root rot.
The Anatomy of a Spiderette: When to Cut and When to Wait
The spider plant utilizes a specialized energy-allocation system. The long stems supporting the plantlets are known as stolons. A stolon serves as a horizontal connection that transports photosynthates and water from the parent plant to the offsets.
Plantlets should remain attached to the stolon while they develop cellular infrastructure. Early removal forces the plantlet to rely on limited energy reserves.
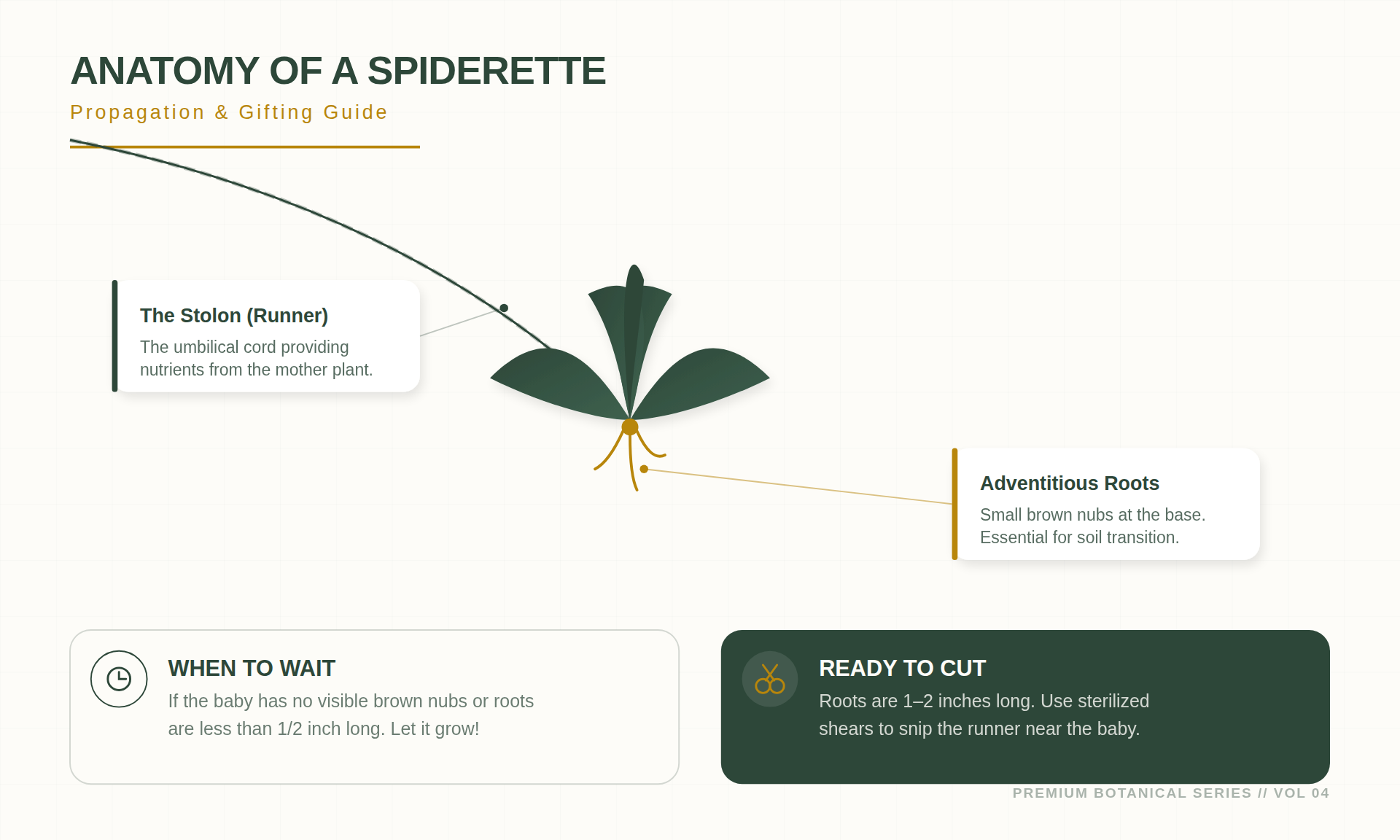
Wait for the development of adventitious roots. These appear as small protrusions at the base of the plantlet. In a natural habitat, these stolons would reach the soil, allowing these nodes to anchor. Removal should only occur when these protrusions are at least 0.5 inches long or visibly swollen. The plantlet should also have four to five well-formed leaves to ensure it has sufficient primordia (specialized root cells) to survive independence.
The Role of Node Development
The node is the point of attachment between the plantlet and the stolon. It contains meristematic activity, where undifferentiated cells can develop into stems, leaves, or roots based on environmental signals. When cutting, leave approximately one inch of the stolon on either side of the plantlet to act as a temporary nutrient buffer during the transition.
The Propagation Protocol: Water, Soil, or Pinning?
There are three primary methods for propagating spider plant offsets, each with different physiological impacts.
Hydroponic Rooting
Placing a plantlet in water typically results in rapid root growth. However, water roots are physiologically distinct from soil roots. They are thicker, more brittle, and optimized for low-oxygen environments. They lack the fine root hairs required to extract moisture from soil particles. Transitioning water-grown plantlets to soil often results in a period of decline as the plant must develop a new root system.
Direct Soil Planting
This method is effective if high humidity is maintained. It requires a light potting medium, such as a 50/50 mix of peat moss (or coco coir) and perlite, to minimize resistance for new root growth.
The Pinning Method
The pinning method involves placing a small pot of moist medium under the plantlet while it remains attached to the parent plant. A U-shaped pin is used to secure the base of the plantlet against the soil.

This allows the plantlet to continue receiving nutrients from the parent plant while developing soil-adapted roots. After approximately three weeks, the stolon can be severed once the plantlet shows resistance to a gentle tug, indicating established roots.
The Critical Root-to-Soil Transition
Transplant shock occurs when the plant’s stomata transpire water faster than the new root system can replace it. This is particularly common in low-humidity environments.
Oxygenation and Soil Structure
Roots require oxygen for cellular respiration. When potting plantlets, the soil should remain loose rather than compacted. A perlite-heavy mix ensures adequate aeration and prevents root suffocation.
For water-rooted plantlets, a “slurry” technique can be used. Gradually add small amounts of soil to the water over two weeks until the medium is entirely moist soil. This allows the plant to develop necessary root hairs. If wilting occurs, increase humidity by using a clear plastic cover, while monitoring for fungal growth.
Pest Quarantine: Preventing Thrips Infestations
Spider plants can host pests such as spider mites and thrips. Their channeled leaves provide shelter for larvae. In dry indoor environments, spider mites are particularly prevalent. New plantlets should undergo a 14-day quarantine and monitoring period using blue sticky traps.
The Smear Test for Pest Detection
To check for pests, wipe the underside of the leaves with a clean white paper towel. Reddish or brown streaks indicate spider mites. Small, dark, moving specks indicate thrips.
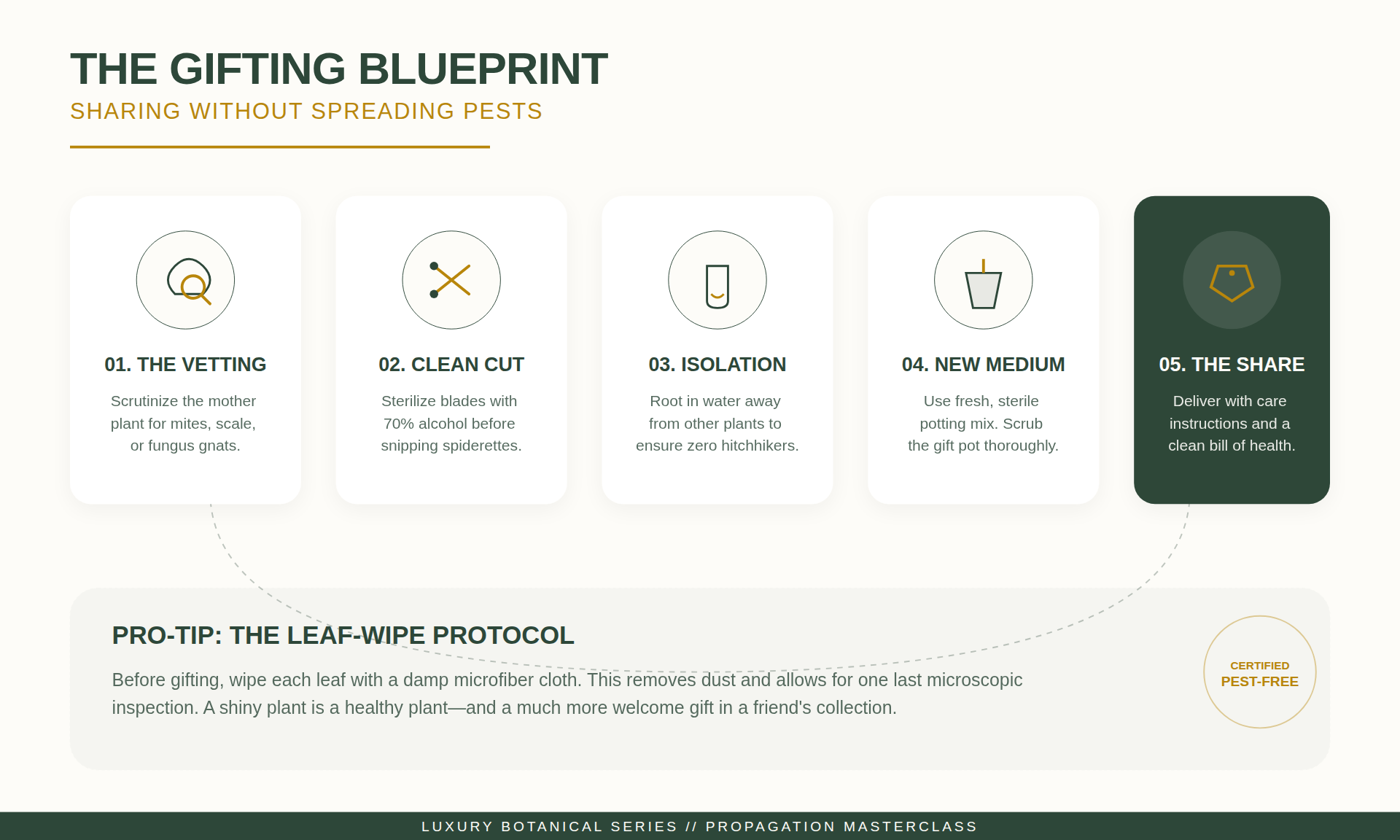
Infested plantlets should not be gifted or moved into general collections. Treatment options include wiping leaves with a 70% isopropyl alcohol solution (diluted) and applying systemic granules to the soil. If the infestation is severe, disposal via composting is recommended to protect other plants.
Gifting Protocols and Pest Prevention
When gifting plantlets, certain standards should be met to ensure the plant’s survival in a new environment.
Terracotta for New Plantlets
Terracotta pots are porous, which facilitates faster soil drying and reduces the risk of overwatering. Ensure the pot has a functional drainage hole to prevent root rot.
Care Documentation
Provide instructions regarding light requirements (bright indirect light) and watering frequency (allow the top inch of soil to dry). Note that spider plants contain compounds that can attract cats; while non-toxic, ingestion can cause mild hallucinogenic effects in felines and physical damage to the plant. A top-dressing of gravel or leca can be used to deter fungus gnats.
Managing Plant Density in Limited Spaces
In confined spaces, it is necessary to limit the number of offsets maintained. A single healthy plant can produce a high volume of plantlets annually.
Nutrient Diversion and Mother Plant Health
Supporting numerous offsets drains the parent plant’s resources. During winter months with lower light levels, the parent plant may sacrifice older leaves to support plantlets, leading to yellowing and reduced vigor.
Maintaining a maximum of five offsets per plant is a practical limit. Removing excess offsets allows the parent plant to redirect energy toward its own root system and foliage.
Vertical Space Management
Macrame hangers or wall-mounted planters can keep stolons clear of floor space. Excess plantlets that cannot be housed or gifted can be composted to recycle nutrients.
Effective plant collection management focuses on the health of individual specimens rather than total quantity.
—
During periods of low indoor humidity, spider plants continue to produce stolons. While other species may struggle, *Chlorophytum comosum* remains active in vegetative propagation.
Guide Overview
- The Anatomy of a Stolon
- The Energy Transfer Mechanism
- Identifying Root Initials
- The Importance of Callousing
- Three Propagation Methods
- Water Rooting
- Soil Rooting
- The Pinning Method
- Tuberous Roots and Moisture Management
- Pest Defense: Thrips and Spider Mites
- Triggering Stolon Production
- Pet Safety and Spider Plants
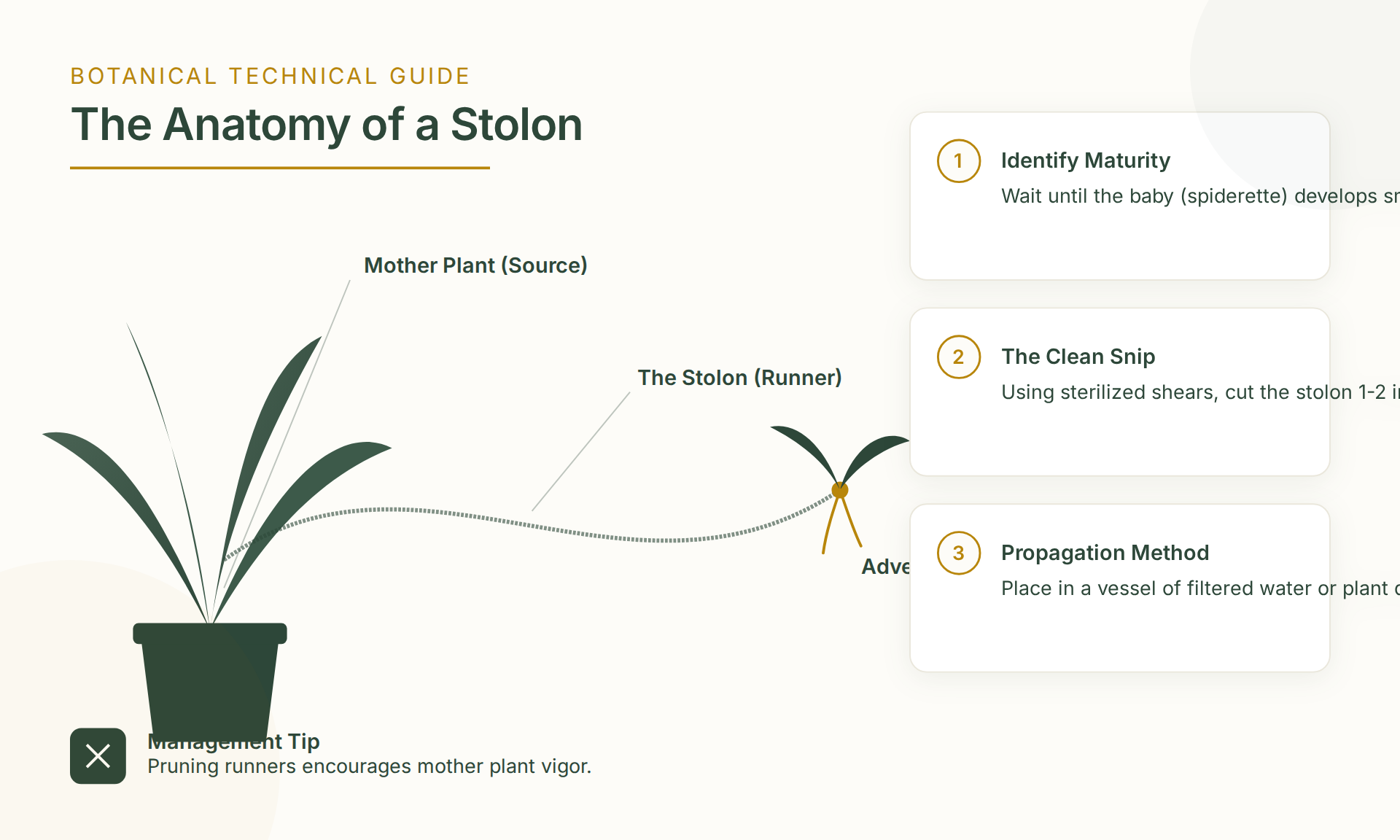
The Anatomy of a Stolon
Stolons are specialized horizontal organs used for vegetative propagation. They function as a vascular highway, transporting nutrients and energy from the parent plant to the developing clone.
In low-light conditions, energy transfer is less efficient. Stolons may become thinner as the plant economizes resources. If light levels are insufficient, the parent plant may abort smaller plantlets. Supplemental LED lighting can support plantlet development during these periods.
The Energy Transfer Mechanism
Stolons contain robust vascular bundles, including xylem and phloem. The phloem transports sugars from the parent (source) to the plantlet (sink). Severing the stolon before the plantlet is self-sufficient can lead to failure. Independence is signaled by a hormonal shift within the plantlet.
Identifying Root Initials
Successful propagation requires identifying “root initials”—small, firm, brownish bumps at the base of the plantlet crown. These are adventitious roots.
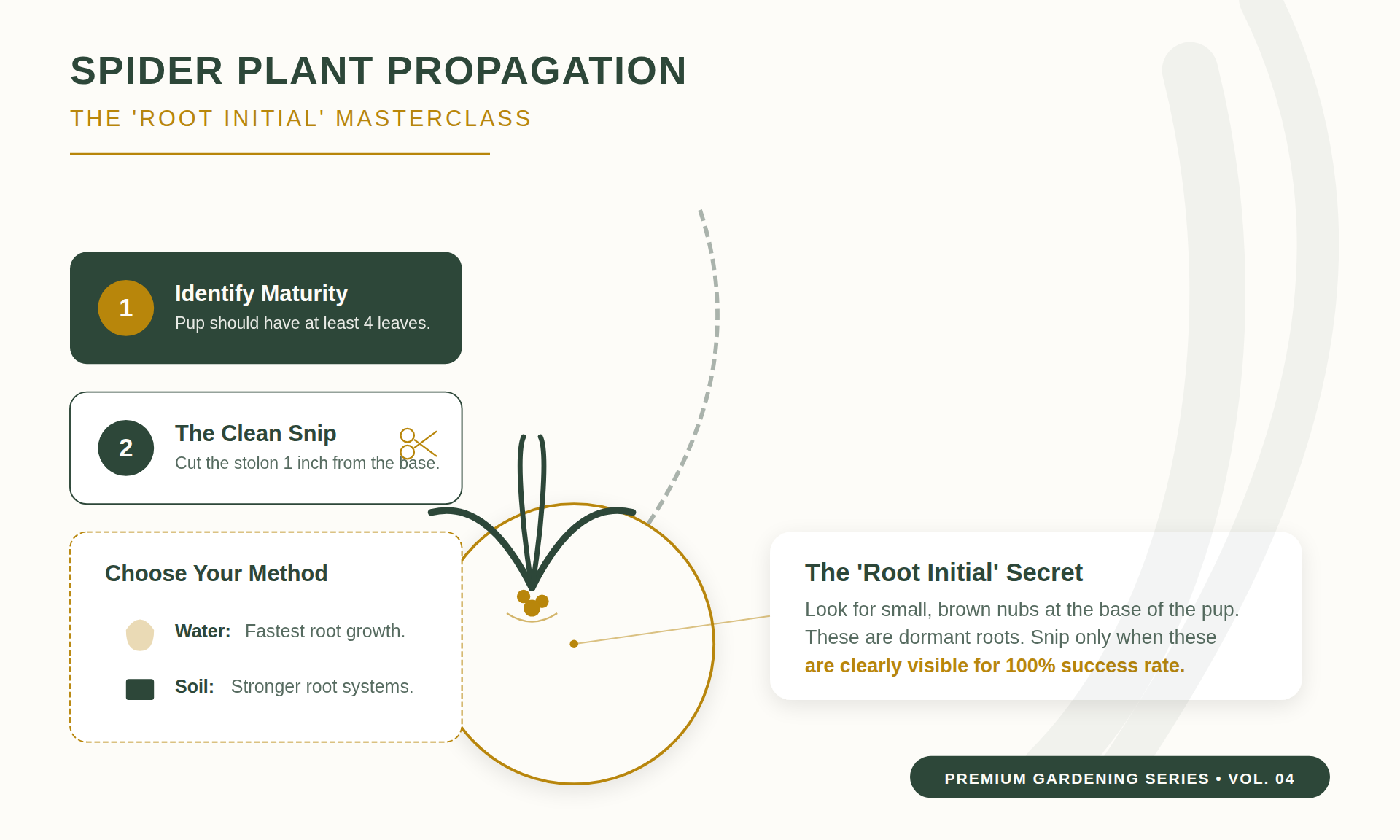
These initials contain meristematic cells that elongate upon contact with moisture. Snipping before these are visible increases the metabolic load on the plantlet.
The Importance of Callousing
Allowing the severed stolon to callous for approximately two hours before planting helps seal the vascular tissue. This prevents pathogens from entering the crown and reduces the risk of rot.
Three Propagation Methods
Water Rooting
Plantlets are placed in water until roots develop. In winter, avoid placing jars directly on cold windowsills to prevent temperature shock to the meristem.
Soil Rooting
Planting directly into a medium is effective for plantlets with visible root initials. A mix of coco coir and perlite provides the necessary oxygen for root development and prevents anaerobic conditions.
The Pinning Method
This method involves securing the plantlet to a separate pot of soil while it remains attached to the parent.

This is the most secure method for variegated or weaker plantlets, as it maintains the nutrient connection until new leaf growth is observed.
Tuberous Roots and Moisture Management
Spider plants (*Asparagaceae* family) develop thick, fleshy, tuberous roots that store water. These roots are susceptible to rot in heavy, peat-based soils that remain damp for extended periods.
To monitor moisture, use a wooden probe to check deep soil levels. A high-performance medium consists of 50% potting soil, 30% perlite, and 20% orchid bark to ensure adequate macropores for root respiration.
Pest Defense: Thrips and Spider Mites
Thrips (order *Thysanoptera*) inhabit the crevices of new growth. Silvering on leaves is a primary indicator of infestation.

A 21-day quarantine is recommended for new plantlets. Spider mites thrive in dry, stagnant air. Regular rinsing of foliage can reduce mite populations. Severely affected plantlets should be removed and disposed of immediately.
Triggering Stolon Production
Stolon production is primarily influenced by the light cycle (photoperiodism). To encourage production, provide 12 hours of bright, indirect light followed by total darkness, mimicking the transition to spring.
Adjusting fertilizer to a higher phosphorus ratio can also signal the plant to produce stolons. Avoid over-fertilizing during dormant periods.
Pet Safety and Spider Plants
Spider plants are non-toxic to pets. However, they contain compounds that are mildly hallucinogenic to cats. Mechanical damage from chewing can affect plant health and aesthetics, and ingestion of fibrous leaves may cause digestive upset. Hanging baskets are an effective way to keep plantlets out of reach of domestic animals.
Regularly cleaning dust from leaves ensures that stomata remain functional for air filtration and plant health.