This article was researched and reviewed by Leo, indoor plant specialist.
Root and Base Decay Analysis: Causes of Structural Failure
A sulfurous odor indicates anaerobic decomposition in Dracaena trifasciata. If the leaves exhibit significant leaning or if a leaf detaches with minimal resistance and a softened texture, the plant is experiencing a systemic failure of vascular integrity. This condition is not merely aesthetic; it represents a breakdown of the plant’s internal transport systems.
Guide Overview
- Root and Base Decay Analysis
- Identifying Irreversible Damage
- CAM Metabolism and Water Requirements
- Succulent Tissue Function
- Remediation: 5 Steps to Resuscitate Saturated Specimens
- Step 1: Root Inspection
- Step 2: Excision of Necrotic Tissue
- Step 3: Hydrogen Peroxide Application
- Step 4: The Callus Period
- Step 5: Replanting in Dry Substrate
- Substrate Engineering: Eliminating Anaerobic Conditions
- Component Breakdown
- The Terracotta Factor
- Differentiating Overwatering from Thermal Cell Collapse
- Differentiating Symptoms
- The Dry-Down Protocol: Maintenance Schedule
- The Substrate Moisture Test
- Seasonal Adjustments and Light Intensity
Intervention against necrosis is required when a snake plant is overwatered. Healthy roots are fibrous and orange or white in color, adapted for arid environments. Chronic saturation displaces soil oxygen, creating anaerobic conditions where pathogens proliferate. The biological shift involves chlorosis (chlorophyll breakdown) followed by the degradation of the rhizome—the underground stem used for energy storage.
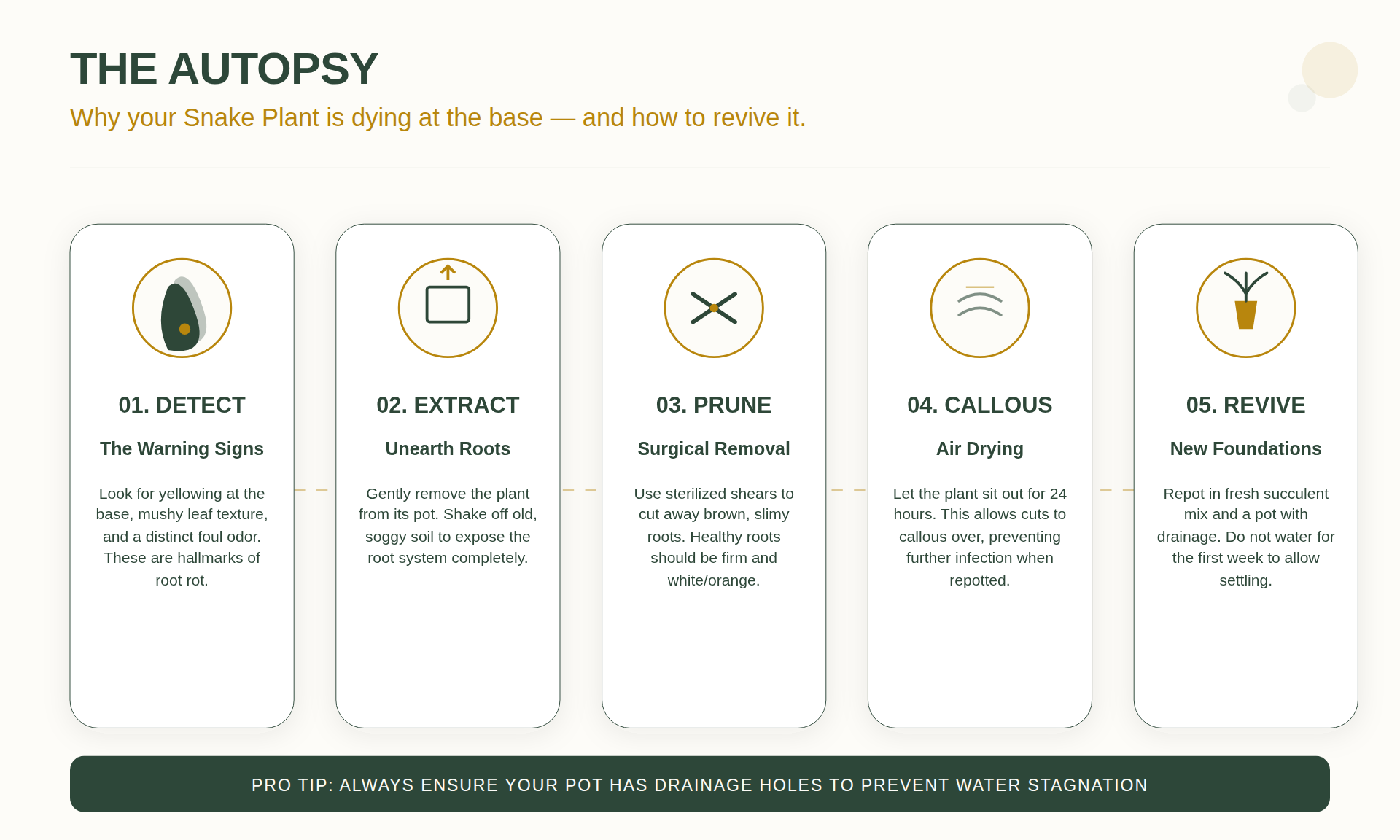
Mushy or translucent-brown tissue at the base indicates dissolved cell walls. This occurs when internal hydrostatic pressure (turgor) exceeds cellular capacity or when anaerobic bacteria digest the plant tissue. Based on observations from 2018, moisture-retaining soils often lead to specimen loss by preventing root respiration. If the rhizome is black and emits a stagnant odor, the plant requires surgical intervention rather than simple desiccation.
Identifying Irreversible Damage
Yellowing leaves are not always terminal. Snake plants may shed outer leaves to reallocate nutrients. However, basal yellowing or the easy removal of the central spear indicates a compromised meristem. The meristem contains undifferentiated cells necessary for growth. If rot reaches this central point, the primary plant is likely non-viable, though healthy tissue may still be salvaged for propagation.
CAM Metabolism and Water Requirements
Snake plants utilize Crassulacean Acid Metabolism (CAM). Unlike C3 or C4 plants that open stomata during the day, CAM plants open stomata at night to absorb CO2 while minimizing water loss through transpiration. They store CO2 as malic acid for processing during daylight hours.
This adaptation results in a low metabolic rate. During periods of low light and temperature, the plant’s metabolic activity decreases further. If water is applied when the stomata are closed and the vascular pumps are inactive, the moisture remains in the substrate, leading to saturation. The plant cannot effectively utilize or transpire excess water under these conditions.
The Role of Succulent Tissue
The leaves function as water storage reservoirs. When soil remains saturated, the plant continues water intake via osmosis. If the storage capacity is exceeded, hydrostatic pressure causes cell wall rupture. This leads to a loss of structural turgidity and subsequent collapse. In natural habitats, these reserves allow survival during extended drought. In domestic settings, frequent irrigation often leads to cellular failure.
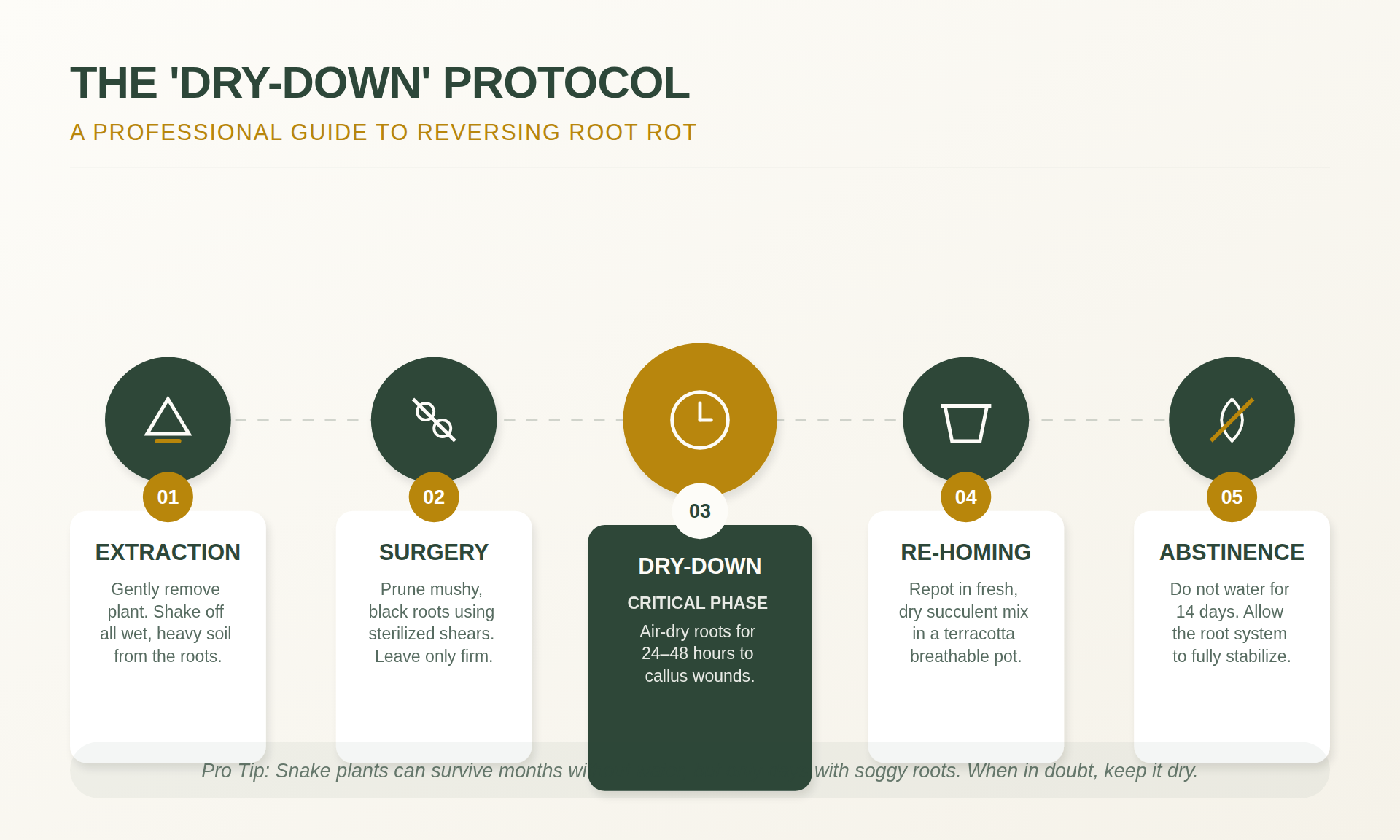
Remediation: 5 Steps to Resuscitate Saturated Specimens
If the root system is saturated, immediate intervention is necessary. Use pruning shears or a sharp blade sterilized with 70% isopropyl alcohol to prevent the transmission of pathogens to healthy tissue.
Step 1: Root Inspection
Remove all existing substrate from the root system. If the soil is compacted, rinse the root ball with room-temperature water to expose the structure. Healthy roots are firm; necrotic roots will exhibit a sliding outer sheath that leaves behind a thin vascular thread. Dead roots must be identified and removed.
Step 2: Excision of Necrotic Tissue
Remove all tissue that lacks firmness. Any hint of softening in the rhizome must be cut back until clean, white or orange tissue is visible. Snake plants can regenerate root systems from a healthy rhizome or leaf cuttings if the necrotic sections are fully removed.

Step 3: Hydrogen Peroxide Application
After excision, soak the remaining healthy tissue in a solution of one part 3% hydrogen peroxide to two parts water for 10–15 minutes. This process neutralizes anaerobic bacteria and provides oxygen to the remaining tissue.
Step 4: The Callus Period
Do not repot the plant immediately. The excision sites must dry and form a callus to prevent secondary infection. Place the plant on a dry surface in a well-ventilated area for 48 to 72 hours. Repotting should only occur once the cut ends are dry and firm to the touch, creating a barrier against soil-borne pathogens.
Step 5: Replanting in Dry Substrate
Use a substrate with minimal moisture. Do not apply water for at least seven days post-repotting. This period of desiccation stimulates auxin production, encouraging the development of new roots.
Substrate Engineering: Eliminating Anaerobic Conditions
Peat-based commercial potting mixes are designed for high water retention, which can lead to compaction and the elimination of macropores. This lack of aeration creates anaerobic conditions. For optimal health, a substrate must allow rapid drainage while maintaining a thin film of moisture on particles.
A 50/50 ratio of organic to inorganic material is recommended to ensure porosity.
Component Breakdown
- 50% Inorganic Grit: Perlite, pumice, or coarse horticultural sand. These components prevent compaction. Pumice is preferred for its weight and stability in the mix.
- 50% Organic Base: Coconut coir or aged pine bark. Coir is preferred over peat as it remains manageable when dry and maintains a neutral pH.
The Terracotta Factor
Unglazed terracotta pots are porous and facilitate moisture evaporation through the vessel walls. This provides a safeguard against overwatering. Conversely, plastic and glazed ceramic pots retain moisture for longer durations, which can be detrimental during periods of low metabolic activity.
Differentiating Overwatering from Thermal Cell Collapse
In winter conditions, cold damage can mimic the symptoms of root rot. Snake plants are tropical and typically require temperatures above 55°F (13°C). At 50°F (10°C), metabolic failure may occur.
Thermal cell collapse results in mushy, translucent leaves. However, cold damage typically affects foliage nearest to cold sources (e.g., windows) while the root system may remain healthy.
Differentiating Symptoms
If leaves become mushy while the substrate is dry, the cause is likely thermal cell collapse. Water within the cells expands upon cooling, rupturing cell walls. Upon warming, the ruptured cells release fluids, creating a softened texture. In this case, do not disturb the roots. Relocate the plant to a warmer area and remove damaged tissue only after the plant has stabilized and the tissue has shriveled.
The Dry-Down Protocol: Maintenance Schedule
Watering frequency should be based on evapotranspiration rates and the substrate saturation point rather than a fixed calendar schedule. Requirements vary significantly based on light intensity and seasonal changes.
The Substrate Moisture Test
To accurately assess moisture, insert a wooden probe to the bottom of the pot. If the probe shows moisture or adhering soil upon removal, irrigation is not required. In winter, the substrate should be completely dry throughout the profile before adding water.
Seasonal Adjustments and Light Intensity
During winter months, light levels often decrease by 50%, requiring a corresponding reduction in watering—often by 75%. Large specimens may only require irrigation every 6 to 8 weeks in low-light conditions.
When irrigation is performed, it should be thorough. Small, frequent applications can lead to surface moisture retention while lower roots remain dry, fostering anaerobic bacteria. The substrate should be fully saturated until water exits the drainage holes, then allowed to dry completely. This cycle oxygenates the root zone and replicates natural rainfall patterns.
Snake plants are adapted for drought resistance rather than saturated environments. Monitoring the rhizome and adjusting for seasonal metabolic shifts is essential for long-term viability.





